Seznamy Atom Particles Charges
Seznamy Atom Particles Charges. Protons have a positive charge while electrons have a negative charge. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … The positive charges equal the negative charges, so the atom has no overall charge;
Prezentováno Antimatter Wikipedia
The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … A proton is a positively charged particle located in the nucleus of an atom. Two of the subatomic particles have electrical charges:Protons have a positive charge while electrons have a negative charge.
Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. Protons have a positive charge while electrons have a negative charge. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. A proton is a positively charged particle located in the nucleus of an atom. The positive charges equal the negative charges, so the atom has no overall charge;

The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. Protons have a positive charge while electrons have a negative charge. There are three subatomic particles: A proton is a positively charged particle located in the nucleus of an atom. Two of the subatomic particles have electrical charges: The positive charges equal the negative charges, so the atom has no overall charge; The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons.. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it …

A proton is a positively charged particle located in the nucleus of an atom... Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. The positive charges equal the negative charges, so the atom has no overall charge; There are three subatomic particles: A proton is a positively charged particle located in the nucleus of an atom. Two of the subatomic particles have electrical charges: Protons have a positive charge while electrons have a negative charge. Protons have a positive charge while electrons have a negative charge.

The positive charges equal the negative charges, so the atom has no overall charge; There are three subatomic particles: An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … The positive charges equal the negative charges, so the atom has no overall charge; A proton is a positively charged particle located in the nucleus of an atom. Two of the subatomic particles have electrical charges: Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons... The positive charges equal the negative charges, so the atom has no overall charge;

The positive charges equal the negative charges, so the atom has no overall charge; Protons have a positive charge while electrons have a negative charge. Two of the subatomic particles have electrical charges: There are three subatomic particles: Two of the subatomic particles have electrical charges:

A proton is a positively charged particle located in the nucleus of an atom. There are three subatomic particles: Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. Two of the subatomic particles have electrical charges: The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. The positive charges equal the negative charges, so the atom has no overall charge; A proton is a positively charged particle located in the nucleus of an atom.. There are three subatomic particles:

An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it ….. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it …

Protons have a positive charge while electrons have a negative charge. Two of the subatomic particles have electrical charges: Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. A proton is a positively charged particle located in the nucleus of an atom. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge.

An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. Protons have a positive charge while electrons have a negative charge. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons... A proton is a positively charged particle located in the nucleus of an atom.

A proton is a positively charged particle located in the nucleus of an atom. Protons have a positive charge while electrons have a negative charge. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. The positive charges equal the negative charges, so the atom has no overall charge; Two of the subatomic particles have electrical charges: There are three subatomic particles:

A proton is a positively charged particle located in the nucleus of an atom. . A proton is a positively charged particle located in the nucleus of an atom.
The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it …. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. A proton is a positively charged particle located in the nucleus of an atom. Protons have a positive charge while electrons have a negative charge. Two of the subatomic particles have electrical charges:.. The positive charges equal the negative charges, so the atom has no overall charge;

An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. A proton is a positively charged particle located in the nucleus of an atom. The positive charges equal the negative charges, so the atom has no overall charge; Protons have a positive charge while electrons have a negative charge. Two of the subatomic particles have electrical charges: Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. There are three subatomic particles:. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it …

A proton is a positively charged particle located in the nucleus of an atom. A proton is a positively charged particle located in the nucleus of an atom. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. There are three subatomic particles: Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles.

The positive charges equal the negative charges, so the atom has no overall charge;. . There are three subatomic particles:

Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. A proton is a positively charged particle located in the nucleus of an atom. There are three subatomic particles: The positive charges equal the negative charges, so the atom has no overall charge; Protons have a positive charge while electrons have a negative charge. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it …

Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles.. There are three subatomic particles: The positive charges equal the negative charges, so the atom has no overall charge; Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. Protons have a positive charge while electrons have a negative charge... Two of the subatomic particles have electrical charges:

Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles.. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. There are three subatomic particles: The positive charges equal the negative charges, so the atom has no overall charge; A proton is a positively charged particle located in the nucleus of an atom. Protons have a positive charge while electrons have a negative charge. Two of the subatomic particles have electrical charges: An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. The positive charges equal the negative charges, so the atom has no overall charge;

Protons have a positive charge while electrons have a negative charge... Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. Protons have a positive charge while electrons have a negative charge. A proton is a positively charged particle located in the nucleus of an atom. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … The positive charges equal the negative charges, so the atom has no overall charge; Two of the subatomic particles have electrical charges: An electron has 11836 times the mass of a proton, but an equal and opposite negative charge.. The positive charges equal the negative charges, so the atom has no overall charge;

An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. A proton is a positively charged particle located in the nucleus of an atom.. Protons have a positive charge while electrons have a negative charge.

A proton is a positively charged particle located in the nucleus of an atom... A proton is a positively charged particle located in the nucleus of an atom. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles.. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge.

Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons... There are three subatomic particles: Two of the subatomic particles have electrical charges: The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it …

The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it …. . Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles.

Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles.. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. A proton is a positively charged particle located in the nucleus of an atom. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it ….. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it …

There are three subatomic particles: Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. A proton is a positively charged particle located in the nucleus of an atom. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. Two of the subatomic particles have electrical charges: The positive charges equal the negative charges, so the atom has no overall charge; Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. There are three subatomic particles: Protons have a positive charge while electrons have a negative charge. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … Protons have a positive charge while electrons have a negative charge.

Two of the subatomic particles have electrical charges: Protons have a positive charge while electrons have a negative charge. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles.
/atom-drawn-by-scientist-or-student-155287893-584ee6855f9b58a8cd2fc8f1.jpg)
An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. Two of the subatomic particles have electrical charges: A proton is a positively charged particle located in the nucleus of an atom. The positive charges equal the negative charges, so the atom has no overall charge; Protons have a positive charge while electrons have a negative charge. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons... An electron has 11836 times the mass of a proton, but an equal and opposite negative charge.

Two of the subatomic particles have electrical charges: The positive charges equal the negative charges, so the atom has no overall charge; Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. A proton is a positively charged particle located in the nucleus of an atom. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. Protons have a positive charge while electrons have a negative charge. Two of the subatomic particles have electrical charges: There are three subatomic particles:. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons.

The positive charges equal the negative charges, so the atom has no overall charge; An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. The positive charges equal the negative charges, so the atom has no overall charge;.. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons.
The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … . A proton is a positively charged particle located in the nucleus of an atom.

Protons have a positive charge while electrons have a negative charge.. Protons have a positive charge while electrons have a negative charge.

Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles... Protons have a positive charge while electrons have a negative charge. The positive charges equal the negative charges, so the atom has no overall charge; Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … There are three subatomic particles: An electron has 11836 times the mass of a proton, but an equal and opposite negative charge... There are three subatomic particles:

Protons have a positive charge while electrons have a negative charge.. A proton is a positively charged particle located in the nucleus of an atom. Protons have a positive charge while electrons have a negative charge. The positive charges equal the negative charges, so the atom has no overall charge; Two of the subatomic particles have electrical charges: The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … There are three subatomic particles: An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles... A proton is a positively charged particle located in the nucleus of an atom.

Protons have a positive charge while electrons have a negative charge. The positive charges equal the negative charges, so the atom has no overall charge; Two of the subatomic particles have electrical charges: There are three subatomic particles: Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. Protons have a positive charge while electrons have a negative charge. A proton is a positively charged particle located in the nucleus of an atom. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … The positive charges equal the negative charges, so the atom has no overall charge;

A proton is a positively charged particle located in the nucleus of an atom... A proton is a positively charged particle located in the nucleus of an atom. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. Two of the subatomic particles have electrical charges: There are three subatomic particles: Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. The positive charges equal the negative charges, so the atom has no overall charge;.. There are three subatomic particles:

Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. There are three subatomic particles: A proton is a positively charged particle located in the nucleus of an atom... An electron has 11836 times the mass of a proton, but an equal and opposite negative charge.

The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it …. There are three subatomic particles: The positive charges equal the negative charges, so the atom has no overall charge; The positive charges equal the negative charges, so the atom has no overall charge;

Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. The positive charges equal the negative charges, so the atom has no overall charge; An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. Protons have a positive charge while electrons have a negative charge. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. Two of the subatomic particles have electrical charges: The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it …. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles.

Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons.. A proton is a positively charged particle located in the nucleus of an atom.. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it …

Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. A proton is a positively charged particle located in the nucleus of an atom. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. Two of the subatomic particles have electrical charges: The positive charges equal the negative charges, so the atom has no overall charge; Protons have a positive charge while electrons have a negative charge. There are three subatomic particles:. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons.

Two of the subatomic particles have electrical charges:.. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. Two of the subatomic particles have electrical charges: Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons... The positive charges equal the negative charges, so the atom has no overall charge;

Two of the subatomic particles have electrical charges: Protons have a positive charge while electrons have a negative charge. A proton is a positively charged particle located in the nucleus of an atom. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. The positive charges equal the negative charges, so the atom has no overall charge;

The positive charges equal the negative charges, so the atom has no overall charge;. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … A proton is a positively charged particle located in the nucleus of an atom. The positive charges equal the negative charges, so the atom has no overall charge; Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles.. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons.

The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … Two of the subatomic particles have electrical charges: Protons have a positive charge while electrons have a negative charge.. Protons have a positive charge while electrons have a negative charge.

An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. The positive charges equal the negative charges, so the atom has no overall charge; Two of the subatomic particles have electrical charges: Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. Protons have a positive charge while electrons have a negative charge. A proton is a positively charged particle located in the nucleus of an atom. There are three subatomic particles: The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … An electron has 11836 times the mass of a proton, but an equal and opposite negative charge.

Protons have a positive charge while electrons have a negative charge. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. A proton is a positively charged particle located in the nucleus of an atom. Protons have a positive charge while electrons have a negative charge. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … Two of the subatomic particles have electrical charges: There are three subatomic particles: The positive charges equal the negative charges, so the atom has no overall charge;.. Protons have a positive charge while electrons have a negative charge.

The positive charges equal the negative charges, so the atom has no overall charge;.. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. Two of the subatomic particles have electrical charges: The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it ….. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it …

Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. A proton is a positively charged particle located in the nucleus of an atom.. Protons have a positive charge while electrons have a negative charge.

Protons have a positive charge while electrons have a negative charge. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … Two of the subatomic particles have electrical charges: Protons have a positive charge while electrons have a negative charge. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. The positive charges equal the negative charges, so the atom has no overall charge;.. Two of the subatomic particles have electrical charges:

The positive charges equal the negative charges, so the atom has no overall charge; Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. A proton is a positively charged particle located in the nucleus of an atom. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. The positive charges equal the negative charges, so the atom has no overall charge; An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it ….. Two of the subatomic particles have electrical charges:

Two of the subatomic particles have electrical charges: The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … The positive charges equal the negative charges, so the atom has no overall charge; A proton is a positively charged particle located in the nucleus of an atom. There are three subatomic particles: Two of the subatomic particles have electrical charges: Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons.. There are three subatomic particles:
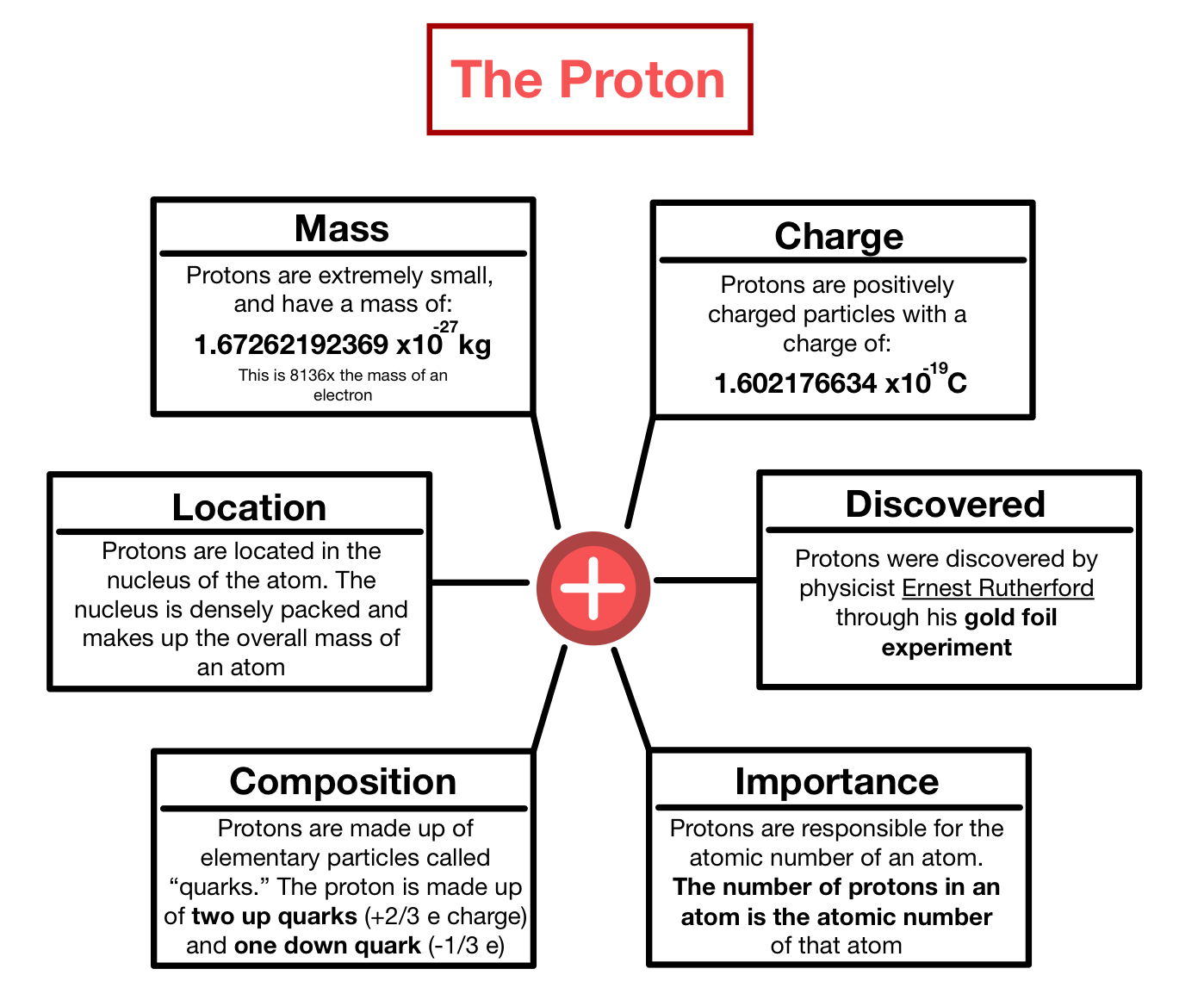
Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles... Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. A proton is a positively charged particle located in the nucleus of an atom. Protons have a positive charge while electrons have a negative charge. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. There are three subatomic particles: The positive charges equal the negative charges, so the atom has no overall charge; Two of the subatomic particles have electrical charges: The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it ….. The positive charges equal the negative charges, so the atom has no overall charge;

A proton is a positively charged particle located in the nucleus of an atom. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons.

An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. A proton is a positively charged particle located in the nucleus of an atom. Protons have a positive charge while electrons have a negative charge. The positive charges equal the negative charges, so the atom has no overall charge;.. A proton is a positively charged particle located in the nucleus of an atom.

Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons.. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge.. Protons have a positive charge while electrons have a negative charge.

The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … The positive charges equal the negative charges, so the atom has no overall charge; The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … Two of the subatomic particles have electrical charges: Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. Protons have a positive charge while electrons have a negative charge. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. There are three subatomic particles: An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. A proton is a positively charged particle located in the nucleus of an atom... The positive charges equal the negative charges, so the atom has no overall charge;

A proton is a positively charged particle located in the nucleus of an atom... . Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles.

Protons have a positive charge while electrons have a negative charge. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles.

The positive charges equal the negative charges, so the atom has no overall charge;.. The positive charges equal the negative charges, so the atom has no overall charge; Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. Two of the subatomic particles have electrical charges: The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … There are three subatomic particles: The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it …

Protons have a positive charge while electrons have a negative charge. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. The positive charges equal the negative charges, so the atom has no overall charge; Two of the subatomic particles have electrical charges: Protons have a positive charge while electrons have a negative charge. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … A proton is a positively charged particle located in the nucleus of an atom. Two of the subatomic particles have electrical charges:

There are three subatomic particles:.. There are three subatomic particles: Two of the subatomic particles have electrical charges: The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … The positive charges equal the negative charges, so the atom has no overall charge; An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles. Protons have a positive charge while electrons have a negative charge. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. A proton is a positively charged particle located in the nucleus of an atom. Sep 18, 2020 · g iven that these particles make up atoms, they are often referred to as subatomic particles.

A proton is a positively charged particle located in the nucleus of an atom. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge. The charge from a proton or electron are of equal strength, therefore if an atom has an equal number of protons and electrons, it … There are three subatomic particles: Two of the subatomic particles have electrical charges: The positive charges equal the negative charges, so the atom has no overall charge; A proton is a positively charged particle located in the nucleus of an atom. Protons have a positive charge while electrons have a negative charge. Jan 19, 2019 · an atom consists of a positively charged nucleus, surrounded by one or more negatively charged particles called electrons. An electron has 11836 times the mass of a proton, but an equal and opposite negative charge.
